ADHD
ADHD and Blood Work: Which Micronutrients Are Actually Missing
11 April 2026 · By Dr. B.J. Huber · 29 min read
Your Brain Needs Raw Materials. And in ADHD, the Right Ones Are Often Missing.
ADHD is usually described as a neurotransmitter problem: not enough dopamine, not enough norepinephrine, disrupted signaling in the prefrontal cortex. That’s accurate. But it’s only half the story.
For your brain to produce dopamine, serotonin, and norepinephrine in the first place, it needs raw materials: iron, zinc, magnesium, B vitamins, omega-3 fatty acids, vitamin D, and specific amino acids like tyrosine and tryptophan. And for those neurotransmitters to function properly, you need a nervous system that isn’t drowning in oxidative stress, and a gut that absorbs nutrients instead of sending inflammatory signals to the brain.
Research over the past decade reveals a consistent pattern: people with ADHD show measurable imbalances across several of these areas. Not everyone, not always the same ones, but significantly more often than the general population. That’s not coincidence. It has biological reasons.
This article gives you the full picture: which blood values and gut parameters are often out of range in ADHD, why that happens, and what you can specifically ask to have tested.
- Serum magnesium looks normal in 67% of ADHD cases despite severe intracellular depletion — you need whole blood magnesium or hair analysis to detect the deficit.
- Iron stores (ferritin) are measurably lower in ADHD across multiple studies, and even “normal” levels below 45 ng/mL may impair dopamine synthesis.
- Dysbiotic microbiome shifts (elevated Bacteroides, reduced Faecalibacterium) damage the gut barrier and trigger neuroinflammation that directly impairs dopamine and serotonin signaling.
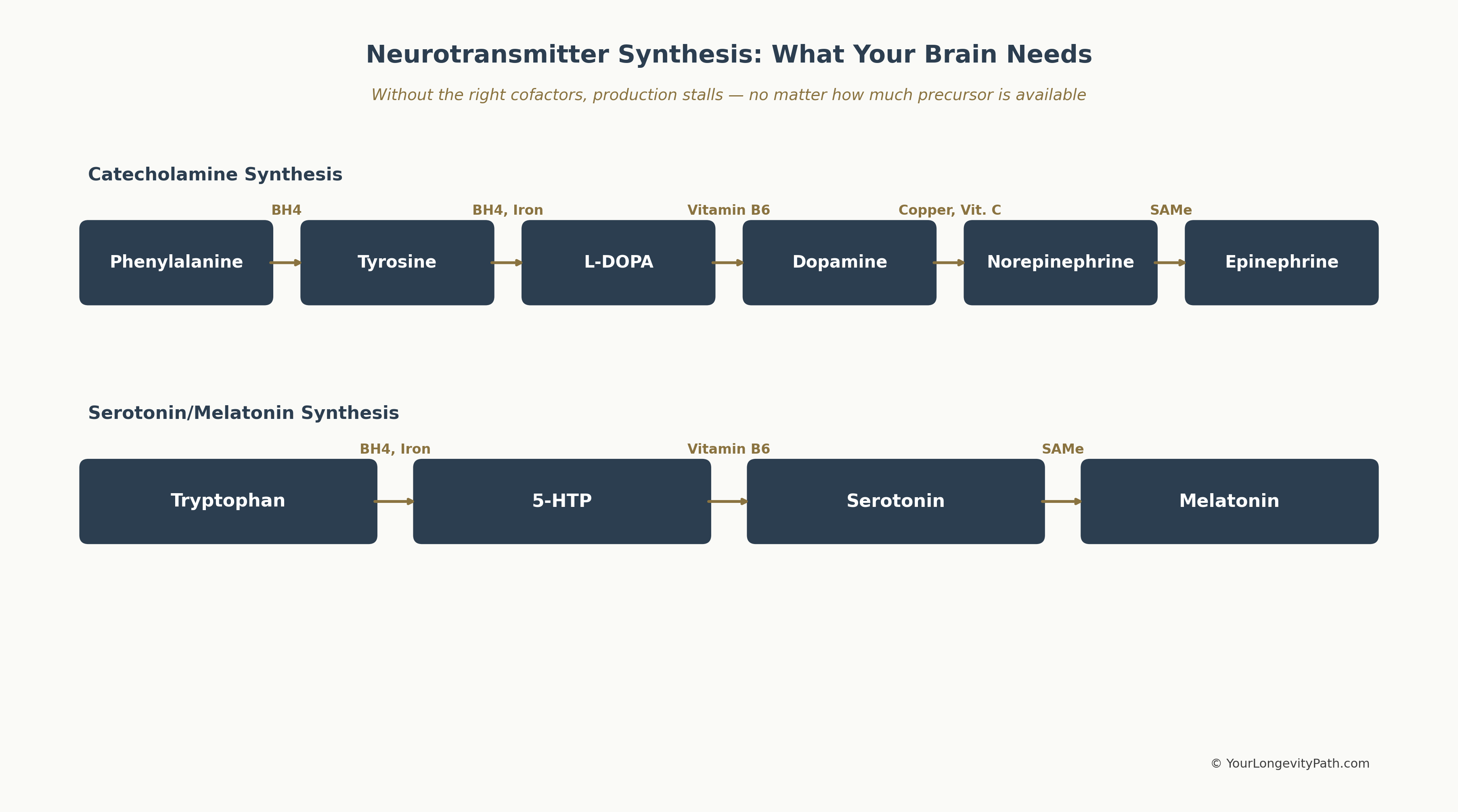 Figure 1: The synthesis pathways of dopamine and serotonin with their respective cofactors. Without the right micronutrients, production stalls at every single step — regardless of how much precursor is available. Own illustration, © YourLongevityPath.com
Figure 1: The synthesis pathways of dopamine and serotonin with their respective cofactors. Without the right micronutrients, production stalls at every single step — regardless of how much precursor is available. Own illustration, © YourLongevityPath.com
The graphic above shows at a glance why a single nutrient deficiency can block the entire neurotransmitter production chain: every arrow requires a specific cofactor. If iron is missing, for example, neither tyrosine can be converted to L-DOPA nor tryptophan to 5-HTP — two completely different neurotransmitter systems are affected simultaneously.
- Your medication can’t manufacture dopamine. Methylphenidate keeps dopamine in the synaptic cleft longer — but without iron, zinc, B vitamins, and magnesium, the raw material is missing.
- Your standard blood panel won’t catch these deficiencies. Serum magnesium can look perfectly normal while your cells are completely depleted. Standard tests fall short here.
- Too much copper, too little zinc — a common ADHD pattern. The result: dopamine gets converted too quickly into stimulating norepinephrine, amplifying inner restlessness.
- Your tryptophan is being misrouted. Instead of making serotonin, it’s broken down via the inflammatory kynurenine pathway — the glutamine/glutamate ratio is shifted by up to 28% (Skalny et al., 2021).
- Your gut is sabotaging your brain. Dysbiosis blocks nutrient absorption and sends inflammatory signals to the brain — massively worsening ADHD symptoms and oxidative stress.
Magnesium: The NMDA Blocker That’s Almost Always Low
Magnesium is the most extensively studied micronutrient in ADHD. A meta-analysis by Effatpanah et al. (2019), pooling seven controlled studies, found significantly lower serum magnesium levels in people with ADHD compared to healthy controls (difference: -0.105 mmol/L, p < 0.013). A second meta-analysis by Huang et al. (2019) confirmed this finding for both serum and hair magnesium.
The numbers become even more striking with intracellular measurements: Kozielec and Starobrat-Hermelin (1997) found magnesium deficiency in 95 percent of children with ADHD examined, most frequently in hair (77.6 percent), followed by red blood cells (58.6 percent) and serum (33.6 percent). This illustrates a critical principle: serum magnesium is a poor indicator of actual magnesium status. If you’re considering magnesium supplementation for ADHD, the specific form matters enormously — see my deep dive into magnesium L-threonate and ADHD.
Why is magnesium low in ADHD?
Three mechanisms interlock. First, stress depletes magnesium. In ADHD, the autonomic nervous system is chronically shifted toward sympathetic dominance, increasing magnesium demand. Pickering et al. (2020) showed this relationship is bidirectional: stress lowers magnesium, and magnesium deficiency amplifies the stress response. Second, stimulant medications like methylphenidate alter the calcium-magnesium ratio and may increase renal magnesium excretion (Schmidt et al., 1994). Third, ADHD-related eating patterns (impulsive food choices, processed foods, reduced appetite on medication) frequently lead to inadequate intake.
What magnesium does in the ADHD brain: It is the natural blocker of the NMDA receptor, protecting against glutamate excitotoxicity — the overstimulation of nerve cells that represents a core problem in ADHD (Sartori et al., 2012).
Recommended tests: Serum magnesium (minimum standard), better: whole blood magnesium or intracellular mineral analysis, since serum values cannot rule out intracellular deficiency.
Iron and Ferritin: The Dopamine Cofactor
Iron is an essential cofactor for tyrosine hydroxylase, the key enzyme in dopamine synthesis. Without sufficient iron, the brain cannot produce enough dopamine — and that is the central problem in ADHD.
A meta-analysis by Cortese et al. (2012) and a systematic review by Wang et al. (2017), published in Scientific Reports, both found the same result: serum ferritin levels are significantly lower in children with ADHD than in healthy controls, with a moderate effect size. Interestingly, serum iron itself was not significantly different, suggesting that iron stores (ferritin) are the more relevant measure.
Why is ferritin low in ADHD?
Research points to several causes. First, genetic variation in iron metabolism plays a role: polymorphisms in genes regulating iron transport may increase risk. Second, intake is often reduced: stimulants suppress appetite, which over months can lead to lower iron consumption. Third, chronic low-grade inflammatory processes, which are more common in ADHD, consume iron.
An important point: ferritin values that fall within the reference range (e.g., 30 ng/mL) can still be functionally too low. Some ADHD researchers consider ferritin levels below 45 ng/mL suboptimal for dopamine synthesis.
RECOMMENDED TESTS: Ferritin (not just hemoglobin), transferrin saturation, CRP (to rule out falsely elevated ferritin from inflammation).
Zinc: Far More Than a Trace Element
Zinc plays a central role in synaptic signaling and serves as a cofactor for over 300 enzymes, including those involved in neurotransmitter synthesis. It modulates dopamine and melatonin receptors and influences GABA signaling.
A meta-analysis by Ghoreishy et al. (2021), published in Scientific Reports with 22 studies (1,280 ADHD subjects, 1,200 controls), found lower blood zinc levels in people with ADHD, although results were statistically heterogeneous. Skalny et al. (2020) also confirmed significantly lower zinc and elevated copper-to-zinc ratios in ADHD.
Why is zinc low in ADHD?
Zinc is excreted at higher rates under stress. The chronic stress levels typical of ADHD increase zinc demand. At the same time, copper competes with zinc for absorption in the gut. When copper is elevated (which is more common in ADHD), zinc absorption is further impaired. Eating habits and appetite suppression from medication also play a role.
Recommended tests: Serum zinc, copper, copper-to-zinc ratio.
Copper: When a Trace Element Is Too High
While most micronutrients in ADHD tend to be too low, copper is often elevated. This isn’t a contradiction — it’s part of the same imbalance.
Copper is a cofactor for dopamine beta-hydroxylase, the enzyme that converts dopamine into norepinephrine. Too much copper means: too much norepinephrine. Excess norepinephrine produces hyperactivity, impulsivity, irritability, and aggression — classic ADHD symptoms.
Skalny et al. (2020) found significantly elevated copper-to-zinc ratios in boys with ADHD, with copper values 12 percent above control levels, primarily driven by a 7 percent drop in serum zinc. Arnold et al. (2005) also showed that children with elevated copper respond more poorly to stimulant medications. The reason: stimulants increase dopamine, which excess copper immediately converts to norepinephrine, worsening symptoms rather than improving them.
Why is copper elevated in ADHD?
The causes are multifactorial. Copper-zinc antagonism: when zinc is low, copper rises relative to it. Estrogen increases copper levels, which could explain why some women with ADHD experience cycle-dependent symptom worsening. Stress and inflammation raise ceruloplasmin, the primary copper transport protein.
Recommended tests: Serum copper, ceruloplasmin, copper-to-zinc ratio.
Vitamin D: Neuroprotective and Almost Always Too Low
Vitamin D is technically not a vitamin but a neurohormone. It regulates over 1,000 genes, including those involved in neuroprotection, immune regulation, and neurotransmitter synthesis.
A meta-analysis by Khoshbakht et al. (2018) with data from over 10,000 children and adolescents found that children with ADHD have significantly lower 25(OH)D levels than healthy controls (weighted mean difference: -6.75 ng/mL). All eight included studies showed this effect. A further analysis of five case-control studies found that low vitamin D levels are associated with a 2.57-fold increased risk of ADHD. Particularly noteworthy: low perinatal vitamin D concentrations were linked to a 40 percent higher risk of ADHD later in life.
Why is vitamin D low in ADHD?
People with ADHD often spend more time indoors (screen time, difficulties with daily organization). Add dietary deficits and, in northern latitudes, generally insufficient sun exposure. There are also hints of genetic variants in vitamin D metabolism that may occur more frequently in ADHD.
Recommended tests: 25(OH)D (25-hydroxyvitamin D). Optimal levels are 40 to 60 ng/mL, not just above the reference threshold of 30 ng/mL.
Omega-3 Fatty Acids: Building Blocks of Cell Membranes
Omega-3 fatty acids, particularly EPA and DHA, are essential for the structure and function of neuronal cell membranes. DHA accounts for roughly 20 percent of brain fatty acids and is critical for the fluidity of synaptic membranes, which in turn affects receptor function.
A meta-analysis by Chang et al. (2018), published in Neuropsychopharmacology, summarized the findings: children and adolescents with ADHD have significantly lower blood levels of EPA and DHA than controls, with a moderate effect size of one-third to one-half of a standard deviation. The omega-6-to-omega-3 ratio was particularly striking: LaChance et al. (2016) found in their meta-analysis that people with ADHD have significantly elevated omega-6-to-omega-3 and AA-to-EPA ratios.
Why are omega-3 levels low in ADHD?
The typical Western diet contains an omega-6-to-omega-3 ratio of roughly 15:1 to 20:1, instead of the evolutionarily adapted 1:1 to 4:1. In ADHD, impulsive eating patterns tend to favor processed, omega-6-rich foods. There is also evidence of polymorphisms in desaturase genes (FADS1, FADS2) that impair the conversion of ALA to EPA and DHA and may be more common in ADHD.
An important note on supplementation: in ADHD, a higher EPA-to-DHA ratio is often recommended (e.g., 2:1 or higher), since EPA has stronger anti-neuroinflammatory effects. The supplementation trials that showed positive results in ADHD typically used EPA-dominant formulations over treatment periods of at least four months.
Recommended tests: Omega-3 Index (target: 8 to 11 percent), AA-to-EPA ratio.
B Vitamins and Homocysteine: The Methylation Cycle
B vitamins — specifically B6 (pyridoxine), B9 (folate), and B12 (cobalamin) — are key players in the methylation cycle, which directly drives the synthesis of dopamine, serotonin, and norepinephrine.
A study by Altun et al. (2018), published in the Zeitschrift für Kinder- und Jugendpsychiatrie, found significantly lower serum levels of B6, B12, and folate in children with ADHD compared to controls. Yektaş et al. (2019) compared serum levels of 48 children with ADHD against healthy controls and found significantly lower B12 values and elevated homocysteine in the ADHD group. B12 levels correlated negatively with hyperactivity and impulsivity symptoms. Homocysteine is a marker for methylation cycle efficiency: when it’s elevated, methylation isn’t working optimally.
Why are B vitamins often suboptimal in ADHD?
This is where genetics enters the picture. Polymorphisms in the MTHFR gene (methylenetetrahydrofolate reductase) reduce the ability to convert folate into its active form, methylfolate. Methylfolate is the key molecule that recycles homocysteine into methionine, driving the entire methylation cycle. Studies suggest that certain MTHFR variants (C677T and A1298C) may be overrepresented in ADHD. On top of that come the usual suspects: appetite loss from stimulants, one-sided diets, and stress-driven increased demand.
Recommended tests: Homocysteine, vitamin B12 (holotranscobalamin is more sensitive than total B12), folate (red blood cell folate is more informative than serum folate), vitamin B6.
Amino Acids: The Direct Building Blocks of Your Neurotransmitters
Amino acids are the immediate precursors of dopamine, serotonin, and norepinephrine. Without them, the brain simply cannot produce these chemical messengers, no matter how good the micronutrient supply is. Research shows that several amino acid pathways are measurably altered in ADHD.
Tyrosine and Phenylalanine: The Dopamine Chain
Phenylalanine is converted to tyrosine, and tyrosine is the direct precursor of L-DOPA, from which dopamine is produced. This is the core cascade in ADHD.
Bornstein et al. (1990) found, in one of the earliest studies on the topic, significantly lower plasma levels of tyrosine, phenylalanine, tryptophan, histidine, and isoleucine in 28 ADD patients compared to 20 controls. The authors interpreted this as evidence of a general defect in amino acid transport or absorption.
More recent research (Bergwerff et al., 2016, 83 ADHD children vs. 72 controls) found no significant differences in blood levels of aromatic amino acids. This sounds contradictory, but makes sense from a biochemical perspective: the problem likely lies less in plasma levels and more in transport across the blood-brain barrier. Tyrosine and tryptophan compete at the LAT1 transporter with branched-chain amino acids (leucine, isoleucine, valine). When these are elevated, fewer dopamine and serotonin precursors reach the brain, even when blood levels appear normal.
An additional factor: the conversion of phenylalanine to tyrosine requires tetrahydrobiopterin (BH4) as a cofactor. BH4 is consumed under oxidative stress, which is chronically elevated in ADHD (as described in the next section). This means: even when enough phenylalanine is available, the conversion to tyrosine can stall.
Tryptophan and the Kynurenine Pathway: When Serotonin Precursors Are Misrouted
Tryptophan is the precursor of serotonin. But only about 5 percent of tryptophan is actually converted to serotonin. The majority (roughly 95 percent) is metabolized through the kynurenine pathway, and this is where a central problem lies in ADHD.
A meta-analysis by Cavaleri et al. (2024), pooling 8 studies with 1,177 participants (707 ADHD, 470 controls), found: people with ADHD have significantly higher kynurenine (SMD = 0.56, p = 0.033) and significantly lower kynurenic acid (SMD = -0.33, p < 0.001).
Why does this matter? Kynurenic acid is neuroprotective: it blocks the NMDA receptor, protecting against glutamate excitotoxicity (similar to magnesium). Less kynurenic acid means less neuroprotection. At the same time, elevated kynurenine can damage nerve cells through neurotoxic metabolites like quinolinic acid (an NMDA receptor agonist).
Aarsland et al. (2015) confirmed this pattern in 133 adults with ADHD vs. 131 controls: significantly lower tryptophan (77.3 vs. 83.6 µmol/L, p = 0.004), lower kynurenic acid (p = 0.03), and lower xanthurenic acid (p = 0.004). The authors also found lower B2 and B6 levels and interpreted this as a nutrient-deficiency-driven shift in kynurenine metabolism.
The gut connection is particularly relevant here: certain gut bacteria (Alistipes, Bacteroides), which are often elevated in ADHD, can consume tryptophan directly in the gut before it even reaches the bloodstream. The result: less tryptophan available for serotonin, less for neuroprotective kynurenic acid, more for neurotoxic metabolites.
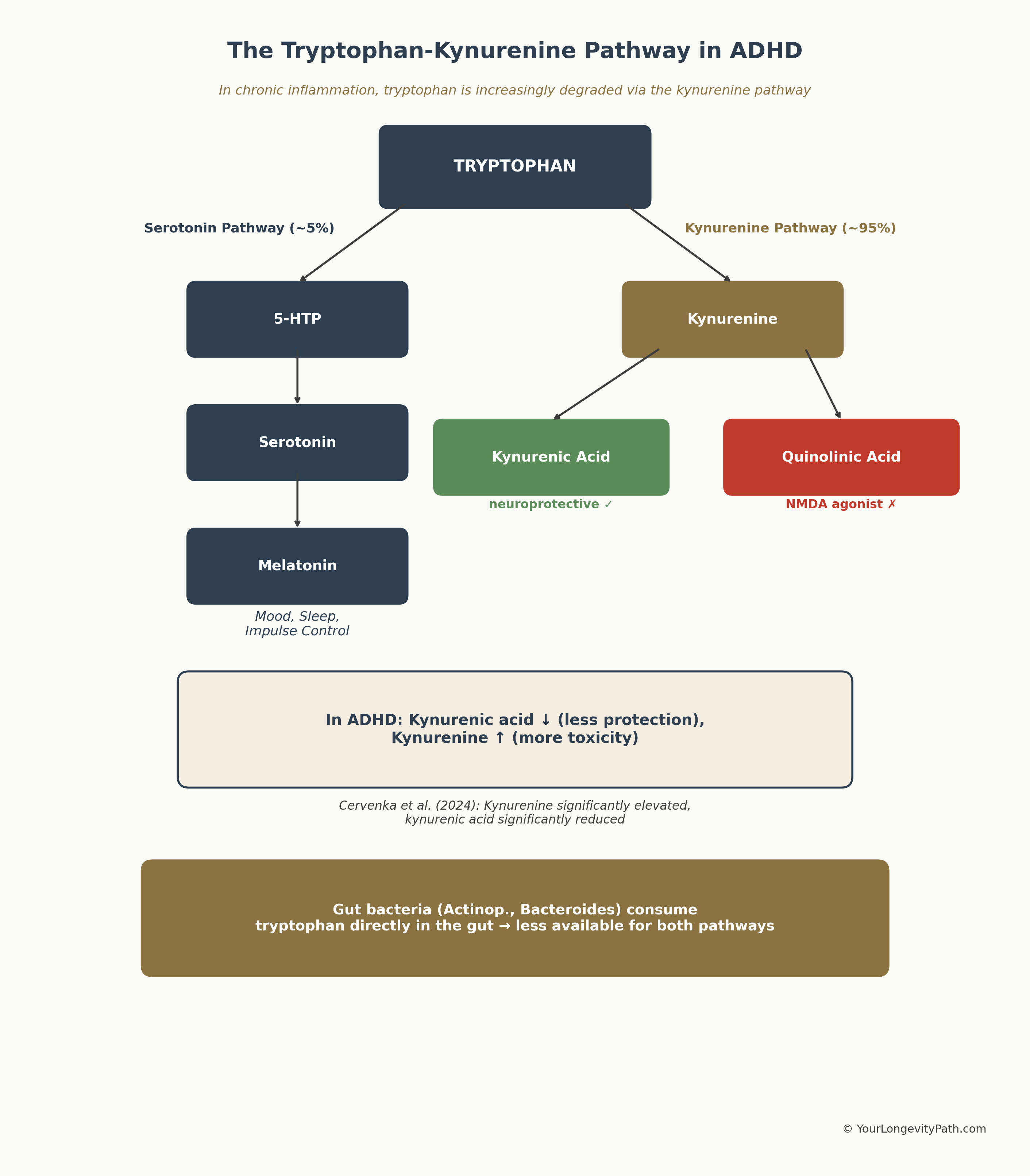 Figure 2: The Tryptophan-Kynurenine Pathway. In chronic inflammation, tryptophan is increasingly degraded via the kynurenine route — less serotonin, less neuroprotection. Own illustration based on Cavaleri et al. (2024), © YourLongevityPath.com
Figure 2: The Tryptophan-Kynurenine Pathway. In chronic inflammation, tryptophan is increasingly degraded via the kynurenine route — less serotonin, less neuroprotection. Own illustration based on Cavaleri et al. (2024), © YourLongevityPath.com
The graphic illustrates why inflammatory processes in ADHD are so problematic: they shift tryptophan metabolism away from the serotonin pathway toward the kynurenine pathway — resulting in less protection and more toxicity.
Recommended tests: Tryptophan (serum), kynurenine, kynurenic acid. These values are measured as part of an extended amino acid analysis and are not included in standard blood panels.
Glutamine and Glutamate: The Excitation Balance
Glutamate is the strongest excitatory neurotransmitter in the brain. Glutamine is its “recycling partner”: in astrocytes, excess glutamate is converted to glutamine to dampen excitation. In ADHD, this balance is disrupted.
Skalny et al. (2021) examined 71 children with ADHD and 31 controls and found: glutamate was 7 percent higher (p = 0.039), glutamine was 10 percent lower (p = 0.024), and the glutamine/glutamate ratio was shifted by 28 percent (p < 0.001). In regression analysis, glutamate levels correlated positively and glutamine levels negatively with ADHD symptom severity.
This connects directly to the magnesium deficit: magnesium blocks the NMDA receptor, which responds to glutamate. When magnesium is lacking and glutamate is simultaneously elevated, a double excitotoxicity arises that damages nerve cells.
Miniksar et al. (2022) confirmed in an independent study (46 children with ADHD, 30 controls) that elevated serum glutamate levels can serve as a biomarker for ADHD, independent of sleep problems.
Recommended tests: Glutamine and glutamate (plasma amino acid profile), glutamine/glutamate ratio.
GABA: The Inhibitory Counterpart
GABA (gamma-aminobutyric acid) is synthesized from glutamate and is the most important inhibitory neurotransmitter. It brakes neuronal overactivity — precisely what is lacking in ADHD.
Several magnetic resonance spectroscopy (MRS) studies show reduced brain GABA concentrations in ADHD: Edden et al. (2012), published in JAMA Psychiatry, found significantly lower GABA levels in children with ADHD. Bollmann et al. (2020) confirmed reduced striatal GABA in unmedicated children with ADHD using high-resolution 7-Tesla MRI.
The paradox: in serum, Miniksar et al. (2022) found elevated GABA levels. This suggests that GABA is being produced but may not efficiently reach the brain or be properly taken up there.
The connection: when glutamate is elevated and GABA is reduced, the excitation/inhibition balance in the brain shifts massively toward excitation. This explains symptoms like sensory overload, impulsivity, and the difficulty of mentally “switching off.”
Histidine: The Underappreciated Alertness Amino Acid
Histidine is the precursor of histamine, a neurotransmitter that regulates alertness, attention, and motivation. Bornstein et al. (1990) found significantly lower plasma histidine levels in ADD patients. Skalny et al. (2021) observed a trend toward 29 percent lower serum histidine levels in children with ADHD (p = 0.081, narrowly non-significant with a small sample size).
The evidence is still thin here, but the mechanism is biochemically plausible: a histidine deficiency could impair histamine production in the brain, thereby contributing to attentional dysfunction.
Recommended tests for the amino acid profile: A comprehensive plasma amino acid profile (with a specialist) can capture tyrosine, phenylalanine, tryptophan, glutamine, glutamate, histidine, and branched-chain amino acids in a single test.
Oxidative Stress: When the Defense Systems Are Overwhelmed
Oxidative stress isn’t a single blood value — it’s a state in which the production of reactive oxygen species (ROS) exceeds the body’s antioxidant capacity. In ADHD, this balance is frequently disrupted.
Ceylan et al. (2012) showed in a controlled study that children with ADHD had significantly lower glutathione peroxidase (GPx) activity and reduced total antioxidant status (TAS). At the same time, malondialdehyde (MDA) levels — a marker of lipid peroxidation (oxidative damage to cell membranes) — were significantly elevated. Bulut et al. (2013) found significantly elevated MDA levels and reduced paraoxonase and arylesterase activity (both antioxidant defense enzymes) in adults with ADHD.
Joseph et al. (2015) summarized the state of research: people with ADHD consistently show reduced levels of antioxidant enzymes (GPx, catalase, SOD) and diminished total antioxidant status (TOS).
Why is oxidative stress elevated in ADHD?
Multiple factors converge. The chronic sympathetic activation typical of ADHD increases ROS production. Micronutrient deficits (zinc, magnesium, selenium) simultaneously weaken antioxidant defense systems, since these minerals are cofactors for antioxidant enzymes. Glutathione, the body’s most important intracellular antioxidant, is consumed faster than it can be replenished under chronic stress. And sleep problems — affecting over 70 percent of people with ADHD — further impair nighttime repair processes.
Recommended tests: Glutathione (GSH/GSSG ratio), MDA (malondialdehyde), total antioxidant status (TAS), SOD activity.
The Gut Microbiome: The Gut-Brain Axis in ADHD
Research on the gut-brain axis in ADHD has gained enormous momentum in recent years. A systematic review by Sukmajaya et al. (2021) and a current review by Mancini et al. (2025) in Neuroscience paint an increasingly clear picture: the composition of the gut microbiome measurably differs between people with ADHD and healthy controls.
What shows up in stool testing:
Reduced diversity. Multiple studies show lower microbial species diversity in ADHD, a pattern also observed in other inflammatory and neuropsychiatric conditions.
Diminished Faecalibacterium abundance. Faecalibacterium prausnitzii is one of the most important butyrate producers in the gut. Butyrate is a short-chain fatty acid that strengthens the gut barrier, inhibits inflammation, and protects the blood-brain barrier. Multiple studies have consistently found reduced Faecalibacterium levels in ADHD, with a negative correlation between Faecalibacterium abundance and ADHD symptom severity.
Altered Bifidobacterium profile. The data on Bifidobacterium is complex: some studies find reduced, others find elevated Bifidobacterium levels in ADHD. A recent study (BMC Microbiology, 2025) found significantly lower abundance of the phylum Actinobacteria (which includes Bifidobacterium) in treatment-naïve children with ADHD.
Elevated Bacteroides and Desulfovibrio. Bacteroides species and Desulfovibriota were elevated in ADHD across multiple studies. Desulfovibrio produces hydrogen sulfide (H₂S), which can damage the gut barrier and promote inflammatory responses.
Why is the microbiome altered in ADHD?
The causes are likely bidirectional. On one hand, ADHD-typical eating patterns (high intake of processed foods, low fiber, irregular meals) promote dysbiosis. On the other, a disrupted gut flora can amplify ADHD symptoms through the gut-brain axis. Mancini et al. (2025) describe a plausible mechanism: dysbiosis leads to increased intestinal permeability (“leaky gut”), allowing bacterial products like lipopolysaccharides (LPS) to enter the bloodstream. These LPS activate peripheral immune cells, which release pro-inflammatory cytokines. Those cytokines can in turn damage the blood-brain barrier and trigger neuroinflammatory processes in the brain that disrupt dopamine and norepinephrine signaling.
One study found elevated plasma levels of lipopolysaccharide-binding protein (LBP) in children with ADHD, an indirect marker for increased intestinal permeability.
Recommended tests: Comprehensive stool analysis with microbiome profiling (16S rRNA sequencing), calprotectin (inflammation marker), zonulin or occludin (markers for intestinal permeability), short-chain fatty acids (butyrate, propionate, acetate), alpha-1-antitrypsin.
Why It All Connects: The Biological Vicious Cycle
These individual deficits don’t exist in isolation. They form a network that amplifies itself.
Magnesium deficiency intensifies the stress response (Sartori et al., 2012), which means more cortisol. More cortisol increases the consumption of zinc and magnesium. Chronic stress simultaneously disrupts the gut microbiome. A disrupted microbiome produces less butyrate, weakening the gut barrier. A permeable gut barrier lets inflammatory mediators through, increasing oxidative stress. Oxidative stress consumes glutathione and antioxidants. And without sufficient antioxidants, cell membranes are damaged, further impairing the absorption of omega-3 fatty acids and other nutrients.
Another example: many people take high doses of vitamin D to correct a deficiency. But to convert vitamin D into its active form, the body absolutely requires magnesium. Taking high doses of vitamin D with a hidden magnesium deficiency depletes your magnesium stores even further, often resulting in a sudden spike of inner restlessness and anxiety.
Stimulant medications can fuel this cycle further: they suppress appetite (less nutrient intake), increase magnesium consumption (Schmidt et al., 1994), and can amplify oxidative stress through sympathetic activation.
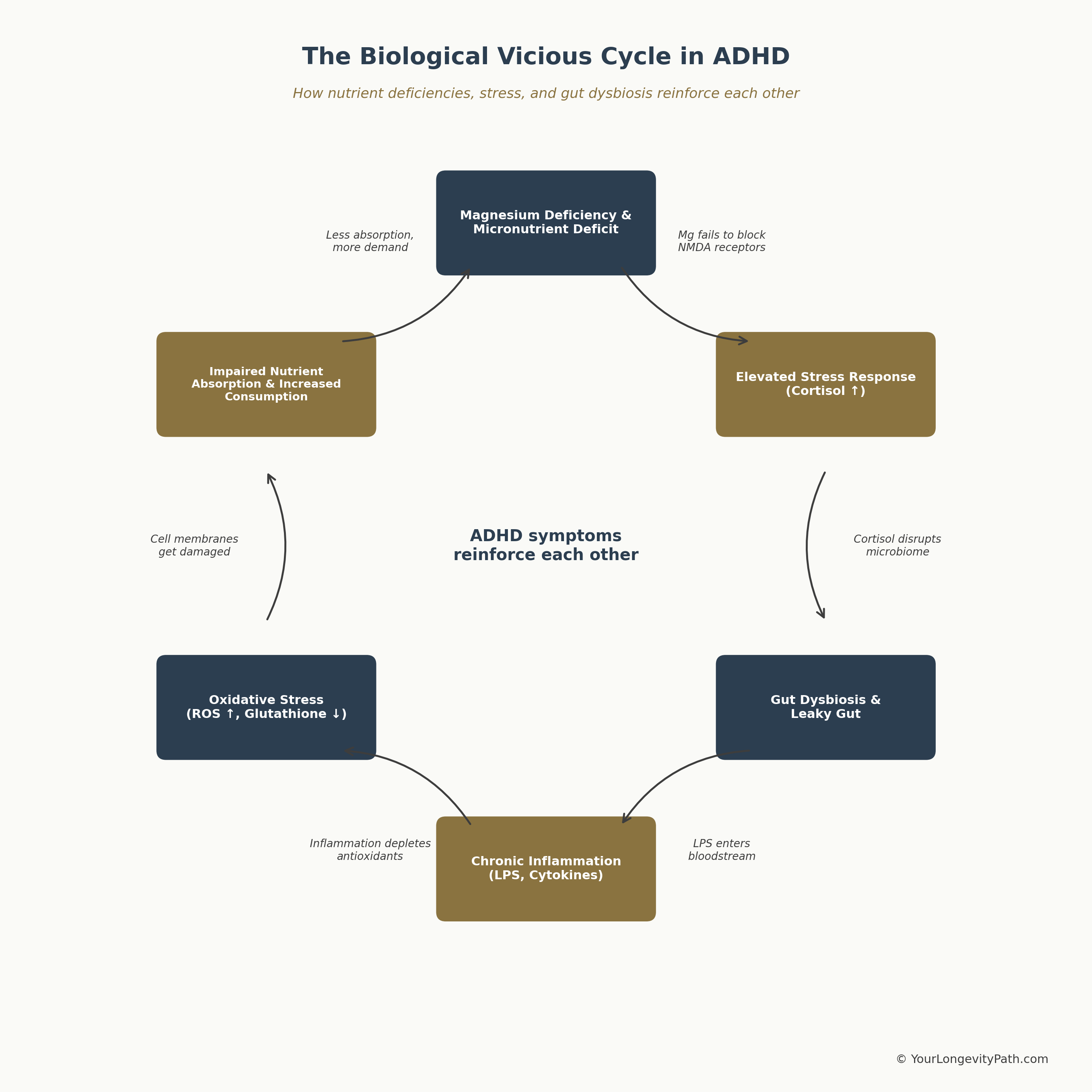 Figure 3: The biological vicious cycle in ADHD. Micronutrient deficiencies, stress, gut dysbiosis, and oxidative stress reinforce each other. Own illustration, © YourLongevityPath.com
Figure 3: The biological vicious cycle in ADHD. Micronutrient deficiencies, stress, gut dysbiosis, and oxidative stress reinforce each other. Own illustration, © YourLongevityPath.com
The graphic shows why isolated interventions often aren’t enough: supplementing magnesium alone while ignoring an inflamed gut won’t break the cycle. An effective approach needs to address multiple points simultaneously.
What You Can Do About It
If you have ADHD and feel like something is missing even though you may already be on medication, here’s a structured approach. The key: not everything at once. Break it into manageable steps.
IMPORTANT: Never supplement blindly. Iron, zinc, and copper in particular should only be taken after a confirmed deficiency on a blood test, since excess levels can be toxic and massively increase oxidative stress. Supplementing iron without a confirmed deficiency can lead to organ damage. The same applies to the copper-zinc ratio: taking zinc in isolation without knowing your copper status can shift the balance in the wrong direction. Always test first, then act.
Prioritize Your Lab Tests (Avoid Overwhelm)
Nobody needs to test everything at once. Break it into two manageable steps:
Step 1: The Baseline Panel (often possible with your GP). Ferritin and transferrin saturation (iron stores), CRP (inflammation marker, to rule out falsely elevated ferritin), 25(OH)D (vitamin D), holotranscobalamin (active vitamin B12, more sensitive than total B12), folate (red blood cell folate is more informative than serum folate), homocysteine (marker for methylation cycle efficiency), and the Omega-3 Index.
Step 2: The Functional Panel (with a specialist). Whole blood magnesium (not serum, since serum values cannot rule out intracellular deficiency), serum zinc, serum copper and the copper-to-zinc ratio, vitamin B6, a plasma amino acid profile (tyrosine, phenylalanine, tryptophan, glutamine, glutamate, histidine), and a comprehensive microbiome stool analysis. For the stool analysis, particularly relevant are: microbiome diversity, Faecalibacterium prausnitzii, short-chain fatty acids (butyrate, propionate, acetate), and inflammation markers such as calprotectin and zonulin.
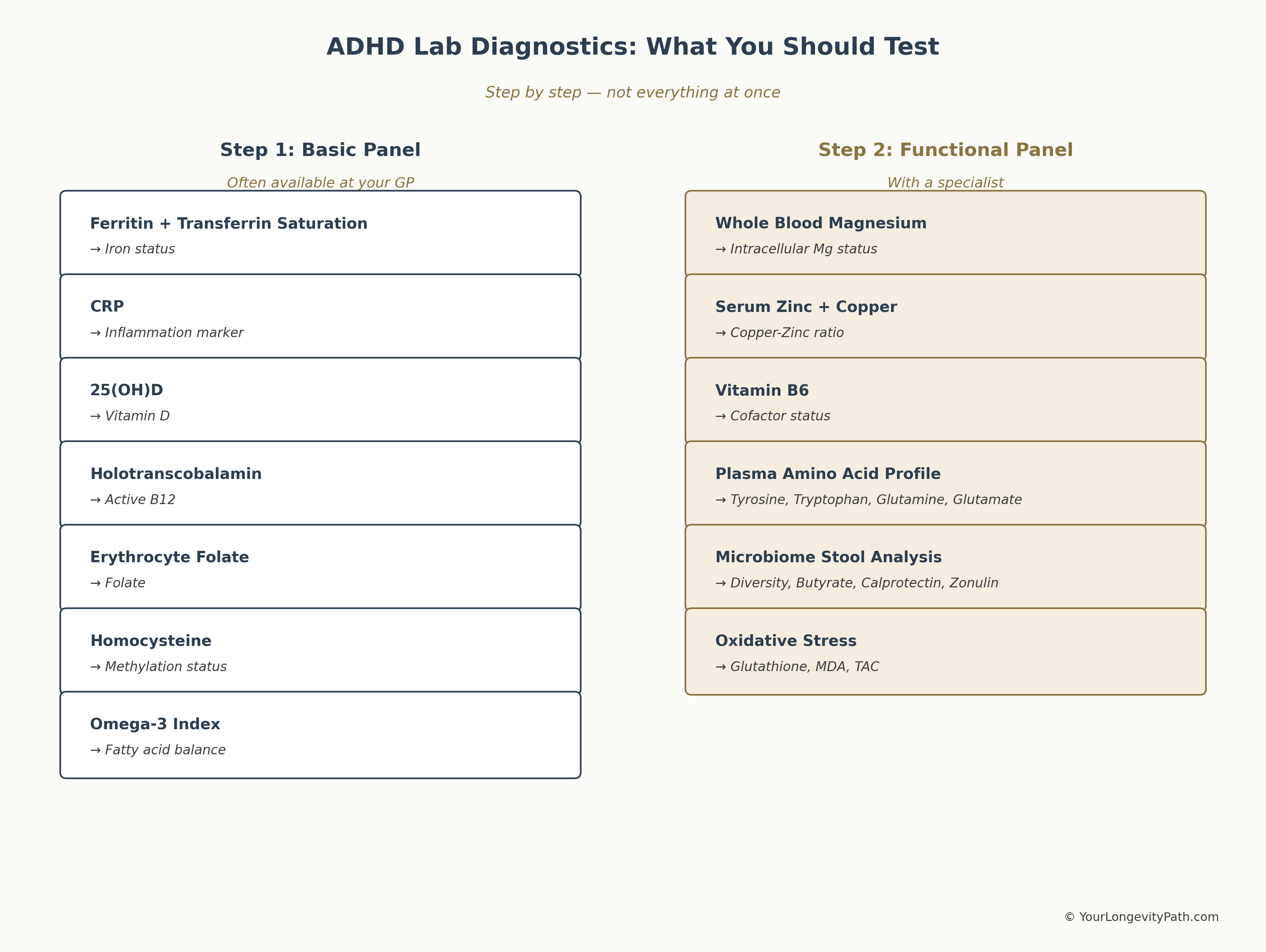 Figure 4: Which tests to run and in what order. Step 1 is often available through your GP, Step 2 requires a functionally trained specialist. Own illustration, © YourLongevityPath.com
Figure 4: Which tests to run and in what order. Step 1 is often available through your GP, Step 2 requires a functionally trained specialist. Own illustration, © YourLongevityPath.com
Discuss the results with someone who can interpret them. Lab values within the reference range are not automatically optimal. The reference range describes where 95 percent of the population falls — not where your values should be for your brain to function at its best. A functionally trained doctor or coach can interpret the numbers in the context of your symptoms and your medication.
Don’t start ten supplements at once. As I explained in my post One step at a time: one change at a time is the approach that works. Identify your biggest deficit and address it specifically.
The Bottom Line
ADHD is not just a neurotransmitter issue. It is a systemic condition where micronutrient deficits, amino acid dysbalances, oxidative stress, and gut-brain axis dysfunction interlock. Research shows measurable imbalances in magnesium, iron (ferritin), zinc, copper, vitamin D, omega-3 fatty acids, B vitamins, amino acids (tryptophan-kynurenine pathway, glutamine/glutamate ratio, GABA), antioxidant capacity, and the composition of the gut microbiome.
That doesn’t mean every person with ADHD has all of these deficits. But it does mean that targeted screening makes sense. Because if you want to optimize a neurological system that’s under constant stress, you have to give it the raw materials it needs. Medications can raise dopamine levels in the synaptic cleft. But they can’t supply the iron that dopamine is made from. They can’t stabilize NMDA receptors when magnesium is missing. And they can’t repair an inflamed gut barrier.
The good news: these parameters are measurable, and many of them are modifiable. Not with a magic pill, but with a structured, evidence-based approach that starts where your body has the greatest need. Understanding these micronutrient basics is part of the broader foundation for understanding longevity and healthspan — just applied specifically to the ADHD brain.
For a deeper dive into magnesium and ADHD, read my post ADHD and Magnesium L-Threonate, where I explain in detail why a specific magnesium form is particularly relevant for the ADHD brain.
Scientific Sources
Aarsland, T. I., Landaas, E. T., Hegvik, T. A., et al. (2015). Serum concentrations of kynurenines in adult patients with attention-deficit hyperactivity disorder (ADHD): a case–control study. Behavioral and Brain Functions, 11, 36. doi: 10.1186/s12993-015-0080-x
Altun, H., Kurutas, E. B., Sahin, N., Gungor, O., & Findikli, E. (2018). The levels of vitamin B6, folate, and vitamin B12 in boys with attention deficit hyperactivity disorder. Zeitschrift für Kinder- und Jugendpsychiatrie und Psychotherapie, 46(5), 396–404. doi: 10.1024/1422-4917/a000559
Arnold, L. E., Bozzolo, H., Hollway, J., et al. (2005). Serum zinc correlates with parent- and teacher-rated inattention in children with attention-deficit/hyperactivity disorder. Journal of Child and Adolescent Psychopharmacology, 15(4), 628–636. doi: 10.1089/cap.2005.15.628
Bergwerff, C. E., Luman, M., Blom, H. J., & Oosterlaan, J. (2016). No Tryptophan, Tyrosine and Phenylalanine Abnormalities in Children with Attention-Deficit/Hyperactivity Disorder. PLOS ONE, 11(3), e0151100. doi: 10.1371/journal.pone.0151100
Bollmann, S., Ghisleni, C., Poil, S. S., et al. (2020). Reduced striatal GABA in unmedicated children with ADHD at 7T. Psychiatry Research: Neuroimaging, 301, 111082. doi: 10.1016/j.pscychresns.2020.111082
Bornstein, R. A., Baker, G. B., Carroll, A., et al. (1990). Plasma amino acids in attention deficit disorder. Psychiatry Research, 33(3), 301–306. doi: 10.1016/0165-1781(90)90046-8
Bulut, M., Selek, S., Bez, Y., et al. (2013). Lipid peroxidation markers in adult attention deficit hyperactivity disorder: New findings for oxidative stress. Psychiatry Research, 209(3), 638–642. doi: 10.1016/j.psychres.2013.02.025
Cavaleri, D., Crocamo, C., Morello, P., Bartoli, F., & Carrà, G. (2024). The kynurenine pathway in attention-deficit/hyperactivity disorder: A systematic review and meta-analysis of blood concentrations of tryptophan and its catabolites. Journal of Clinical Medicine, 13(2), 583. doi: 10.3390/jcm13020583
Ceylan, M. F., Sener, S., Bayraktar, A. C., & Kavutcu, M. (2012). Changes in oxidative stress and cellular immunity serum markers in attention-deficit/hyperactivity disorder. Psychiatry and Clinical Neurosciences, 66(3), 220–226. doi: 10.1111/j.1440-1819.2012.02330.x
Chang, J. P. C., Su, K. P., Mondelli, V., & Pariante, C. M. (2018). Omega-3 polyunsaturated fatty acids in youths with attention deficit hyperactivity disorder: A systematic review and meta-analysis. Neuropsychopharmacology, 43(3), 534–545. doi: 10.1038/npp.2017.160
Cortese, S., Angriman, M., Lecendreux, M., & Konofal, E. (2012). Iron and attention deficit/hyperactivity disorder: What is the empirical evidence so far? Expert Review of Neurotherapeutics, 12(10), 1227–1240. doi: 10.1586/ern.12.116
Edden, R. A. E., Crocetti, D., Zhu, H., Gilbert, D. L., & Mostofsky, S. H. (2012). Reduced GABA concentration in attention-deficit/hyperactivity disorder. Archives of General Psychiatry (now JAMA Psychiatry), 69(7), 750–753. doi: 10.1001/archgenpsychiatry.2011.2280
Effatpanah, M., Rezaei, M., Effatpanah, H., et al. (2019). Magnesium status and attention deficit hyperactivity disorder (ADHD): A meta-analysis. Psychiatry Research, 274, 228–234. doi: 10.1016/j.psychres.2019.02.040
Ghoreishy, S. M., Ebrahimi Mousavi, S., Asoudeh, F., & Mohammadi, H. (2021). Zinc status in attention-deficit/hyperactivity disorder: A systematic review and meta-analysis of observational studies. Scientific Reports, 11, 14612. doi: 10.1038/s41598-021-94124-5
Huang, Y. H., Zeng, B. Y., Li, D. J., et al. (2019). Significantly lower serum and hair magnesium levels in children with attention deficit hyperactivity disorder than controls: A systematic review and meta-analysis. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 90, 134–141. doi: 10.1016/j.pnpbp.2018.11.012
Schmidt, M. E., Kruesi, M. J. P., Elia, J., Borcherding, B. G., Elin, R. J., Hosseini, J. M., McFarlin, K. E., & Hamburger, S. D. (1994). Effect of dextroamphetamine and methylphenidate on calcium and magnesium concentration in hyperactive boys. Psychiatry Research, 54(2), 199–210. doi: 10.1016/0165-1781(94)90006-X
Joseph, N., Zhang-James, Y., Perl, A., & Faraone, S. V. (2015). Oxidative stress and ADHD: A meta-analysis. Journal of Attention Disorders, 19(11), 915–924. doi: 10.1177/1087054713510354
Khoshbakht, Y., Bidaki, R., & Salehi-Abargouei, A. (2018). Vitamin D status and attention deficit hyperactivity disorder: A systematic review and meta-analysis of observational studies. Advances in Nutrition, 9(1), 9–20. doi: 10.1093/advances/nmx002
Kozielec, T., & Starobrat-Hermelin, B. (1997). Assessment of magnesium levels in children with attention deficit hyperactivity disorder (ADHD). Magnesium Research, 10(2), 143–148. PMID: 9368235
LaChance, L., McKenzie, K., Taylor, V. H., & Vigod, S. N. (2016). Omega-6 to omega-3 fatty acid ratio in patients with ADHD: A meta-analysis. Journal of the Canadian Academy of Child and Adolescent Psychiatry, 25(2), 87–96. PMID: 27274744
Mancini, V. O., et al. (2025). Gut dysbiosis as a driver of neuroinflammation in attention-deficit/hyperactivity disorder: A review of current evidence. Neuroscience, 569, 298–312. doi: 10.1016/j.neuroscience.2025.01.024
Miniksar, D. Y., Özdem, S., & Guseinova, N. (2022). GABA, glutamate and homocysteine serum levels and their association with aggression, impulsivity and inattention in children with ADHD. Psychiatry Research, 311, 114473. doi: 10.1016/j.psychres.2022.114473
Pickering, G., Mazur, A., Trousselard, M., et al. (2020). Magnesium status and stress: The vicious circle concept revisited. Nutrients, 12(12), 3672. doi: 10.3390/nu12123672
Sartori, S. B., Whittle, N., Hetzenauer, A., & Singewald, N. (2012). Magnesium deficiency induces anxiety and HPA axis dysregulation: Modulation by therapeutic drug treatment. Neuropharmacology, 62(1), 304–312. doi: 10.1016/j.neuropharm.2011.07.027
Skalny, A. V., Mazaletskaya, A. L., Ajsuvakova, O. P., et al. (2021). Serum amino acid spectrum in children with attention deficit hyperactivity disorder (ADHD). Journal of Trace Elements in Medicine and Biology, 65, 126717. doi: 10.1016/j.jtemb.2021.126717
Sukmajaya, A. C., Lusida, M. I., Soetjipto, S., & Setiawati, Y. (2021). Systematic review of gut microbiota and attention-deficit hyperactivity disorder (ADHD). Annals of General Psychiatry, 20(1), 12. doi: 10.1186/s12991-021-00330-w
Skalny, A. V., Mazaletskaya, A. L., Ajsuvakova, O. P., Bjørklund, G., Skalnaya, M. G., Chao, J. C.-J., Chernova, L. N., Shakieva, R. A., Kopylov, P. Yu., & Tinkov, A. A. (2020). Serum zinc, copper, zinc-to-copper ratio, and other essential elements and minerals in children with attention deficit/hyperactivity disorder (ADHD). Journal of Trace Elements in Medicine and Biology, 58, 126445. doi: 10.1016/j.jtemb.2019.126445
Wang, Y., Huang, L., Zhang, L., et al. (2017). Peripheral iron levels in children with attention-deficit hyperactivity disorder: A systematic review and meta-analysis. Scientific Reports, 7(1), 14029. doi: 10.1038/s41598-017-19096-x
Yektaş, Ç., Alpay, M., & Tufan, A. E. (2019). Comparison of serum B12, folate and homocysteine concentrations in children with autism spectrum disorder or attention deficit hyperactivity disorder and healthy controls. Neuropsychiatric Disease and Treatment, 15, 2213–2219. doi: 10.2147/NDT.S212361
What blood tests should you get for ADHD?
Ferritin, zinc, whole-blood magnesium, vitamin D, omega-3 index, and homocysteine. Standard panels rarely include these — you need to request them specifically.
Can nutrient deficiencies make ADHD symptoms worse?
Yes. Magnesium, iron, zinc, and omega-3 are directly involved in dopamine and noradrenaline synthesis. A deficiency can measurably impair focus, impulse control, and sensory filtering.
Why does gut health matter for ADHD?
Over 90% of serotonin is produced in the gut. Dysbiosis impairs nutrient absorption and raises inflammatory markers that directly affect brain function and neurotransmitter balance.
This article is for informational purposes only and does not replace professional medical advice, diagnosis, or treatment. If you have health concerns, please consult a qualified healthcare professional.